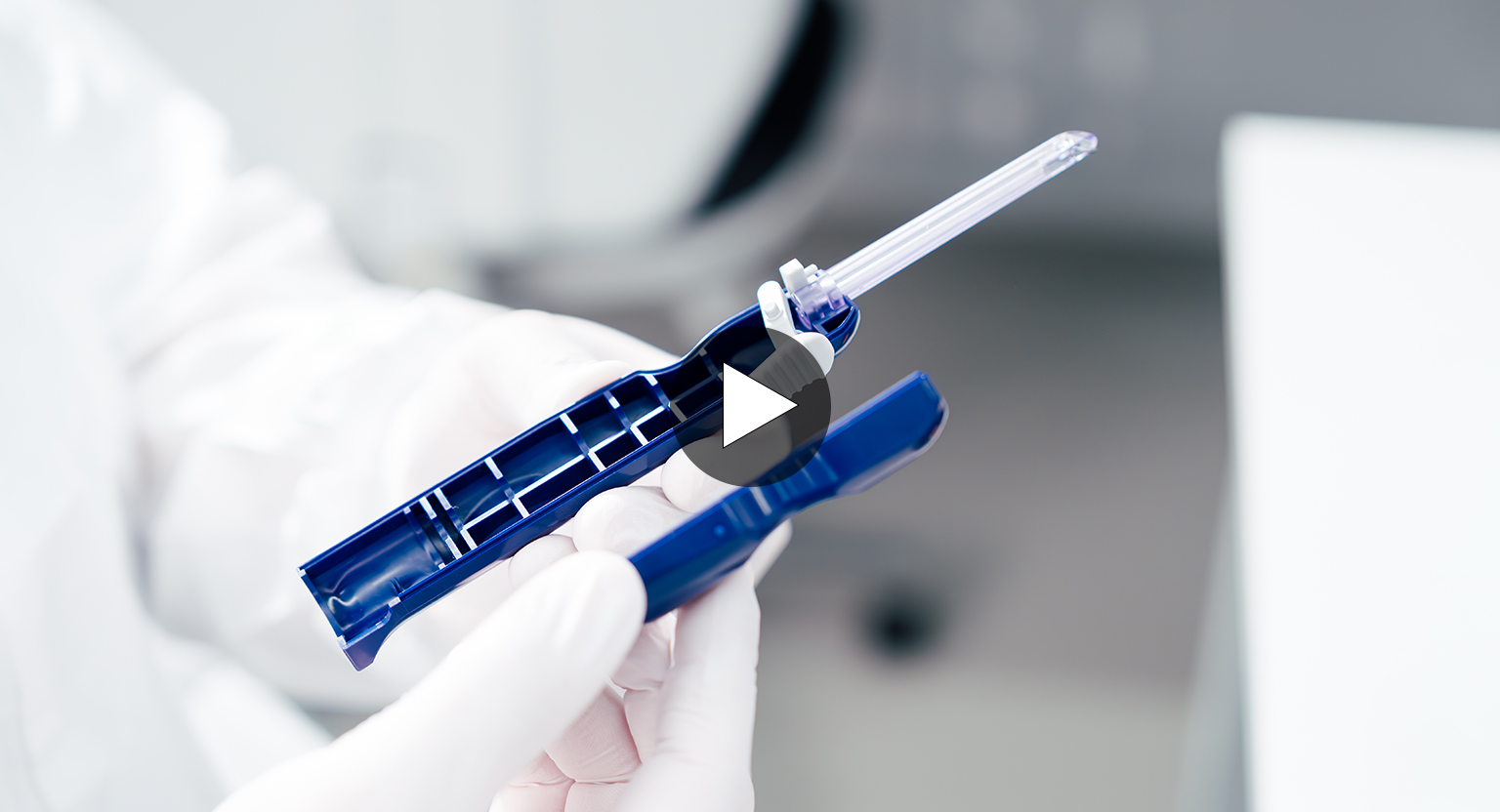
Injection Molding and Cleanroom Manufacturing of Single-Use Instruments
Trend in Medical Technology: Single-Use Instruments
Single-use Instruments have long become an important product segment in medical technology. This trend is primarily driven by increasing requirements for hygiene, patient safety, availability, and reproducible clinical processes. Disposable instruments eliminate the need for reprocessing and reduce associated risks and efforts, making them attractive for many applications.
New opportunities enabled by Single-use Instruments
Single-use instruments enable:
- More cost-efficient care: No reprocessing, sterilization or reverse logistics required
- Improved patient safety: Reduced risk of cross-contamination, reprocessing errors or material-related wear
- New product designs: Economical implementation of function-integrated plastic components, delicate guiding elements or hybrid parts with metal or sensor inserts
Co-Engineering, DFM & Process Validation
One of the most underestimated success factors in medical technology projects lies before the start of serial production. Decisions made in early project phases have a major impact on cost optimization, iteration cycles, and scrap rates.
In Co-engineering and Design for Manufacturing (DFM), the following aspects are evaluated early on:
- Feasibility
- Material selection (ABS, PC compounds, etc.)
- Tolerances
- Demolding behavior and tool design
- Joining and assembly concepts
- Particle and contamination risks
- Testing and validation strategies
- FMEA, PFMEA
- Prototyping
- Packaging
MDR and FDA regulations impose high requirements on the quality and safety of medical devices. At the same time, ISO 13485 defines a quality management standard across the entire product lifecycle and serves as a reference for certification bodies during audits. Manufacturers must demonstrate that processes consistently deliver safe results under defined conditions. Therefore, process validation for manufacturing, joining and cleanroom packaging steps is of critical importance.
Injection Molding Production: From Granules to a Single-use Instrument
In injection molding, quality depends on maintaining tolerances, reproducibly creating surfaces and controlling downstream processes.
Critical processes are specifically carried out under controlled conditions, such as:
- Assembly
- Visual inspection
- Final cleaning
- Packaging
Injection Molding in Cleanroom Production Areas

Injection Molding and Cleanroom Manufacturing at Gsell
Processing high-performance polymers places significant demands on machines, tools and materials. Gsell’s injection molding infrastructure is specifically designed for medical technology requirements.
This helps reduce risks already during production:
- Minimization of particle sources
- Reduced material losses
- Compliance with tight tolerances
Material Selection: Polymers as a Key Success Factor
In medical technology, polymers often need to meet several critical requirements:
- Biocompatibility
- Mechanical stability
- Temperature resistance
- Sterilizability
- Reliable processability
Suitability depends on the application, product class, sterilization method, geometry and production volume. For single-use projects, ABS and PC compounds are particularly attractive due to their mechanical performance, thermal stability and biocompatibility.
Depending on the project, customer-supplied materials or specially qualified compounds can also be processed. This can be crucial when existing material approvals and defined regulatory documentation chains must be maintained.

Granules: Application Example with Dark Blue ABS Compounds
Insert Molding – Overmolding of Inserts
Insert Molding involves placing pre-manufactured components such as metal parts (e.g., blades), threaded inserts, sensors or other functional elements into the mold and directly overmolding them with plastic.
The benefit of this method is functional integration. Instead of assembling multiple components afterward, hybrid parts are created with clearly defined interfaces and often improved reproducibility. This reduces assembly effort and increases product stability. This approach is often both technically and economically advantageous for guiding elements, handle components, fixation features or instrument parts with metallic functions.
Cleanroom Marking: Pad Printing
In the medical environment, pad printing is far more than visual finishing. It enables functional and regulatory identification, such as scales, batch references, position markings or branding elements.
Pad Printing Application on a Single-use Instrument
Key factors in the cleanroom:
- Low particle generation
- Suitable inks and materials
- Validated processes
- Durable and legible marking
- Quality and traceability concepts
Pad Printing result on a Single-use Instrument
Pre-Assembly of Subassemblies: Polymer meets metal
Single-use instruments often consist of functional assemblies rather than a single polymer component. During pre-assembly, multiple components are combined.
Typical assembly services:
- Blade integration into handles
- Threaded inserts
- Snap-fit and locking mechanisms
- Guiding elements
- Mechanical fixation
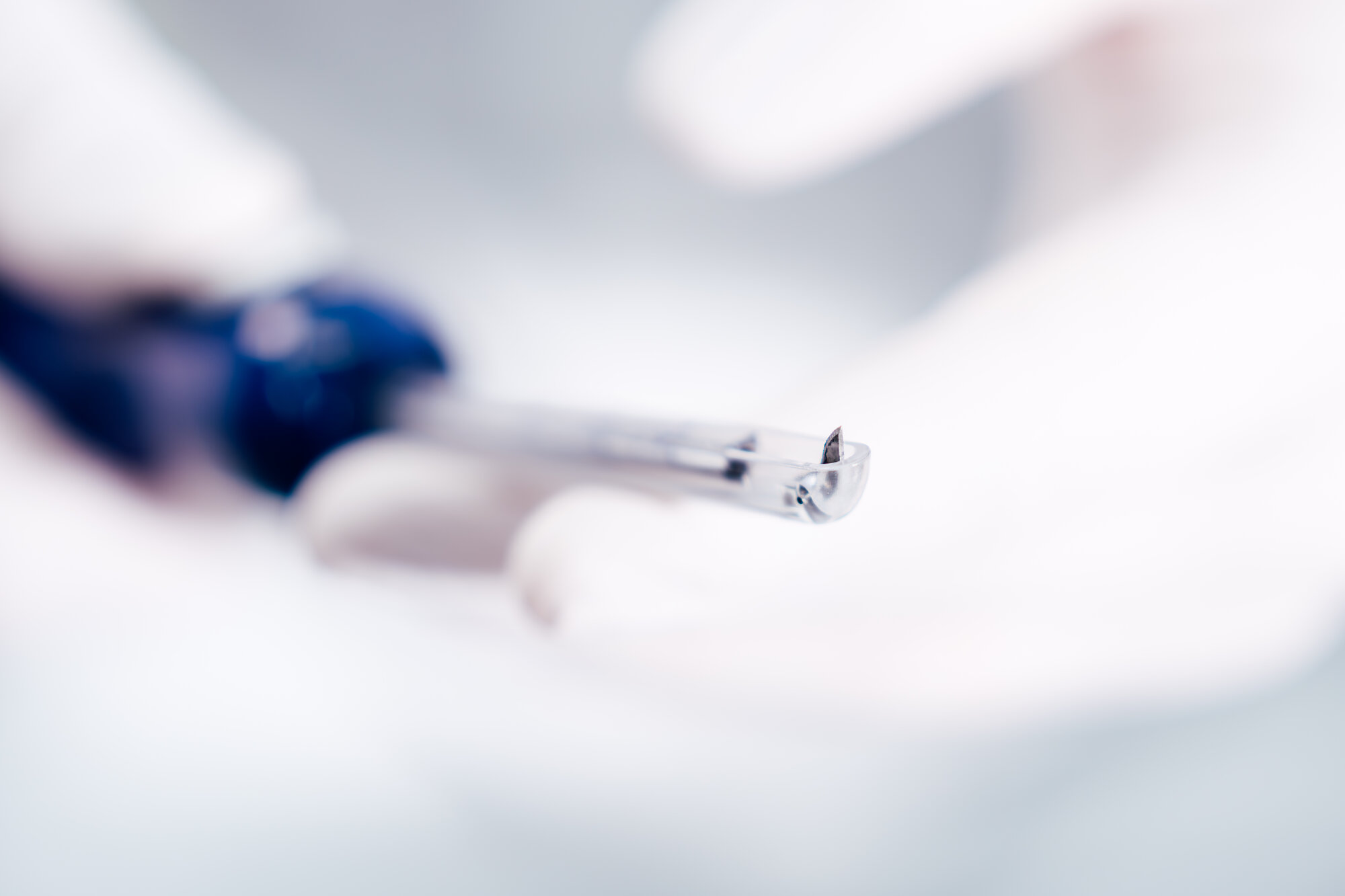
Pre-Assembly of a metal blade
Variations in tolerances, surface properties or press-fit behavior can increase assembly time, raise scrap rates or affect final product performance. Aligning design, injection molding, and pre-assembly leads to both technical and economic advantages.
Ultrasonic Welding: Clean Joining Without Additives
Ultrasonic welding is an established method in medical technology for quickly and cleanly joining plastics without additional materials. This is particularly advantageous for single-use instruments since no adhesives are required.
Ultrasonic welding in Cleanroom
The process can be standardized and validated effectively. With proper design, it produces strong, low-particle joints with defined seam quality.
Ultrasonic welding is suitable for:
- Housings
- Covers
- Fluid-handling assemblies
Cleanroom Packaging
The final step is often not the simplest but one of the most critical: cleanroom packaging. In medical technology, it is not a downstream afterthought but an integral part of the overall process.
Packaging systems and processes are highly relevant for medical devices and must ensure product protection, usability and process reliability.
Typical steps include:
- Visual inspection
- Placement into pouches or blisters
- Sealing
- Preparation for sterilization
What are the benefits of integrated Injection Molding and Cleanroom Manufacturing?
✔ End-to-End process chain
Fewer interfaces and higher efficiency accelerate decision-making and improve the feasibility of complex products.
✔ Validated processes & traceability
Regulatory-compliant validated processes and complete documentation reduce risks and simplify audits.
✔ Faster time to market
Coordinated processes shorten development timelines.
✔ Consistent quality & scalability
Defined processes ensure reproducible quality while enabling scalability from small to large production volumes.
✔ Material expertise
Reliable processing of standard plastics through to customer-specific compounds.
FAQ: Injection Molding and Cleanroom Manufacturing of Single-use Instruments
Why is injection molding ideal for single-use instruments?
The process enables high volumes, excellent reproducibility and comparatively low unit costs at scale. Functional features can be directly integrated into the component, reducing assembly effort.
When is cleanroom manufacturing required for single-use instruments?
This depends on the product’s intended use, risk assessment, contamination requirements and downstream processes. Not all production steps must occur in a cleanroom, but critical steps such as assembly, final cleaning or packaging are often performed under controlled conditions. This is especially important for products with high cleanliness, sterility or regulatory requirements.
Which materials are best suited for injection molding and cleanroom manufacturing of single-use instruments?
Various thermoplastics, such as ABS and PC compounds. The optimal choice depends on sterilization requirements, mechanical stress, temperature profile, geometry and regulatory documentation.
What role does process validation play?
A central one. In medtech projects, processes must be documented as traceable and reproducible. This applies not only to injection molding but also to joining, marking, and packaging processes. Under MDR, FDA and ISO 13485, this traceability is especially critical.
How quickly can a new manufacturing process be developed?
Depending on complexity, requirements and validation effort, it can take several weeks to a few months. Simple components can be industrialized faster than complex assemblies with hybrid functions, marking, assembly and validated packaging.
Planning a Single-use Instrument or looking to optimize existing solutions?
Whether simple plastic components, thick-walled parts, delicate inserts or high-volume production with tight tolerances: our injection molding specialists are ready to take on any challenge.
Contact us – we will find a solution.

Raphael Schwegler
Senior Sales & Project Manager
raphael.schwegler@gsell.ch
 Close
Close